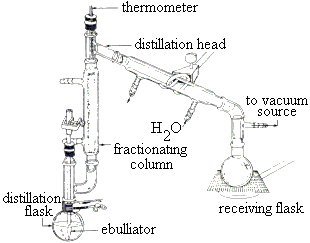
Air is filtered to remove dust, and then cooled in stages until. distillates collected over a narrow temperature range are known as fractions. Question: Nitrogen and oxygen can be separated by the fractional distillation of liquid air.the liquid runs into a collecting vessel.any gas that makes it to the top of the fractioning column enters the condenser where it is changed to a liquid.the liquids which condense in the column drip back into the flask.the vapour condense when they reach a part of the column that is below the temperature of their boiling point.their vapours rise through a column which is hot at the bottom and cooler at the top.the liquids boil at their boiling point.It is useful for separating ethanol from a mixture of ethanol and water, and for separating crude oil into different products such as petrol, diesel and kerosene. Immiscible liquids are ones that can’t – like oil and water.įractional distillation separates miscible liquids that have different boiling points. keeping foods fresh, flushing out dangerous vapours from oil tankers.Miscible liquids are ones that can mix together – like water and ethanol. Manufacture of oxygen using liquefaction and fractional distillation of air.Uses of oxygen and of liquid nitrogen (two examples in each case).Unit C2: Further Chemical Reactions, Rates and Equilibrium, Calculations and Organic Chemistry.2.9.3 demonstrate knowledge and understanding of using nitrogen as a coolant and in food packaging.2.9.1 recall the composition of the gases in the atmosphere: about 78% nitrogen about 21% oxygen about 0.03–0.04% carbon dioxide about 1% argon small proportions of other noble gases and varying proportions of water vapour.Unit 2: Further Chemical Reactions, Rates and Equilibrium, Calculations and Organic Chemistry.It is useful for separating ethanol from a mixture of ethanol and water, and for separating different. Oxygen is removed from the mixture by passing it over hot copper. 
Under the proper conditions, a fraction containing roughly 60 noble gases, 30 oxygen, and 10 nitrogen can be obtained from liquid air. Water vapour and carbon dioxide are removed because they solidify at low. The heavier noble gases argon, krypton, and xenon are obtained by the fractional distillation of liquid air. Air is filtered to remove dust and other solids, water vapour and carbon dioxide. The liquid air gets separated depending on the boiling point of the gas. The separated gases are obtained by the receiver ends from the fractional distillation column. C Oxygen gas is produced if the electrode is made from carbon. Fractional distillation is used to separate different liquids from a mixture of liquids. Many gases are obtained in commercial quantities through a process known as fractional distillation of liquefied air. Correct option is B) The components of liquid air on passing through a fractional distillation column separate based on boiling point. How is xenon obtain from It is obtained by fractional distillation of liquid air. Gases are separated from liquid air by fractional distillation. (i) the air as a source of nitrogen, oxygen, neon and argon Most of the gases in air, Nitrogen, Oxygen, Argon and others are obtained by fractional distillation of liquid air.(e) the present composition of the atmosphere and how the composition of the atmosphere has changed over geological time.Unit 1: CHEMICAL SUBSTANCES, REACTIONS and ESSENTIAL RESOURCES.Unit 2: ENERGY, RATE AND CHEMISTRY OF CARBON COMPOUNDS.Industrial processes are designed to maximise profit and minimise the impact on the environment.8.26a Describe the composition of today’s atmosphere.5.2.2 How bonding and structure are related to the properties of substances.5.2 Bonding, structure and the properties of matter.Students should be able to: predict the states of substances at different temperatures given appropriate data.4.2.2 How bonding and structure are related to the properties of substances.4.2 Bonding, structure and the properties of matter Distillation is a widely used method for separating mixtures based on differences in the conditions required to change the phase of components of the mixture.RSC Yusuf Hamied Inspirational Science Programme.Introductory maths for higher education.The separation happens when the mixture is heated at a certain temperature where fractions of the mixture start to vaporize. The process involves repeated distillations and condensations and the mixture is usually separated into component parts. Since it is a homogeneous mixture, we need special separation techniques. Fractional distillation is a type of distillation which involves the separation of miscible liquids. It consists of gases like nitrogen, oxygen, carbon dioxide, argon, etc. The physics of restoration and conservation Step 1:Conversion of air into liquid air Step 2:Fractional Distillation Separation Techniques: Obtaining Gases from Air We know that air is a homogeneous mixture of gases.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
